A Nurse Is Reviewing the Arteriol Blood Gas for a Client Who Has Respritory Acidosis
An introduction to acid-base balance in health and disease
Normal cell metabolism depends on the maintenance of blood pH within very narrow limits (vii.35-7.45).
Even relatively balmy excursions outside this normal pH range can have deleterious furnishings, including reduced oxygen commitment to tissues, electrolyte disturbances and changes in centre muscle contractility; survival is rare if claret pH falls below 6.eight or rises above 7.8.
The problem for the torso is that normal metabolism is associated with continuous production of hydrogen ions (H+) and carbon dioxide (COtwo), both of which tend to reduce pH. The machinery which overcomes this trouble and serves to maintain normal blood pH (i.e. preserve acid-base homeostasis) is a complex synergy of action involving chemical buffers in claret, the red cells (erythrocytes), which circulate in blood, and the function of three organs: lungs, kidneys and encephalon.
Before explaining how these 5 elements contribute to the overall maintenance of blood pH, it would exist helpful to quickly review some basic concepts.
What is an acid, what is a base and what is pH?
An acrid is a substance which releases hydrogen ions (H+) on dissociation in solution.
For example: Hydrochloric acid (HCl) dissociates to hydrogen ions and chloride ions
HCl![]() H+ + Cl-
H+ + Cl-
Carbonic acid (HiiCO3) dissociates to hydrogen ions and bicarbonate ions
HiiCO3 ![]() H+ + HCO3 –
H+ + HCO3 –
We distinguish between strong acids like hydrochloric acid and weak acids like carbonic acid. The departure is that strong acids dissociate more than weak acids. Consequently the hydrogen ion concentration of a strong acid is much college than that of a weak acid.
A base of operations is a substance which in solution accepts hydrogen ions.
For instance, the base of operations bicarbonate (HCO3 –) accepts hydrogen ions to form carbonic acid:
HCO3 – + H+ ![]() HiiCO3
HiiCO3
pH is a scale of 0-14 of acerbity and alkalinity. Pure water has a pH of 7 and is neutral (neither acidic nor alkaline). pH to a higher place seven is alkali metal and below 7 acidic. Thus the pH of blood (vii.35-seven.45) is slightly alkaline although in clinical medicine the term alkalosis is, perhaps confusingly, reserved for blood pH greater than 7.45 and the term acidosis is reserved for blood pH less than 7.35.
pH is a measure of hydrogen ion concentration (H+). The two are related co-ordinate to the following equation:
pH = - log10 [H+]
where [H+] is the concentration of hydrogen ions in moles per liter (mol/L)
From this equation
pH 7.4 = H+ concentration of twoscore nmol/L
pH 7.0 = H+ concentration of 100 nmol/L
pH 6.0 = H+ concentration of 1000 nmol/L
It is clear that:
- the two parameters alter inversely; as hydrogen ion concentration increases, pH falls
- due to the logarithmic relationship, a big change in hydrogen ion concentration is actually a small alter in pH. For example, doubling the hydrogen ion concentration causes pH to autumn past but 0.three
What is a buffer? – the bicarbonate buffer arrangement
Buffers are chemicals in solution which minimize the change in pH which occurs when acids are added by 'mopping upwardly' hydrogen ions. A buffer is a solution of a weak acrid and its conjugate base of operations. In blood, the principle buffer system is the weak acid, carbonic acrid (HtwoCO3) and its cohabit base, bicarbonate (HCO3 –). To explain how this system minimizes changes in pH, suppose we add a stiff acid, e.k. HCl, to the bicarbonate buffer:
The acid will dissociate, releasing hydrogen ions:
HCl ![]() H+ + Cl–
H+ + Cl–
The bicarbonate buffer then 'absorbs' the hydrogen ions, forming carbonic acid in the process:
HCO3 – + H+ ![]() H2CO3 (carbonic acrid)
H2CO3 (carbonic acrid)
The important point is that because the hydrogen ions from HCl accept been incorporated into the weak carbonic acid, which does non dissociate as hands, the total number of hydrogen ions in solution and therefore the pH do not change as much as would have occurred in the absence of the buffer.
Although a buffer greatly minimizes pH change, it does not eliminate it because even a weak acid (similar carbonic acid) dissociates to some extent. The pH of a buffer solution is a role of the relative concentrations of the weak acid and its cohabit base.
pH = six.1 + log ([HCO3 –] / [H2COiii])
Where [HCO3 –] = concentration of bicarbonate
[H2CO three] = concentration of carbonic acid
This relationship, known as the Henderson-Hasselbalch equation, shows that pH is governed by the ratio of base of operations (HCO3 –) concentration to acid (HtwoCO3) concentration.
As hydrogen ions are added to the bicarbonate buffer:
H+ + HCO3 –![]() H2COiii
H2COiii
bicarbonate (base of operations) is consumed (concentration decreases) and carbonic acid is produced (concentration increases). If hydrogen ions continue to exist added, all bicarbonate would eventually be consumed (converted to carbonic acid) and at that place would be no buffering effect – pH would then fall sharply if more acid were added.
However, if carbonic acid could be continuously removed from the system and bicarbonate constantly regenerated, then the buffering capacity and therefore pH could be maintained despite continued addition of hydrogen ions.
As volition become clear with more detail of the physiology of acid-base of operations balance, that is, in effect, what happens in the trunk. In essence, the lungs ensure removal of carbonic acid (as carbon dioxide) and the kidneys ensure continuous regeneration of bicarbonate.
This function of the lungs is dependent on a singular characteristic of the bicarbonate buffering system and that is the ability of carbonic acid to be converted to carbon dioxide and water.
The following equation outlines the human relationship of all elements of the bicarbonate buffering system as it operates in the body
H+ + HCO3 –![]() HtwoCOiii
HtwoCOiii![]() H2O + COtwo
H2O + COtwo
It is of import to note that the reactions are reversible. Management is dependent on the relative concentration of each element. So that, for example, a rise in carbon dioxide concentration forces reaction to the left with increased formation of carbonic acid and ultimately hydrogen ions.
This explains the acidic potential of carbon dioxide and brings us to the important contribution that the lungs and blood-red cells make to overall acrid-base of operations balance.
Lung office, ship of COii and acrid-base balance
A constant amount of CO2 in blood, essential for normal acid-base of operations remainder, reflects a remainder between that produced equally a effect of tissue cell metabolism and that excreted past the lungs in expired air.
By varying the rate at which carbon dioxide is excreted, the lungs regulate the carbon dioxide content of blood. The sequence of events from carbon dioxide production in the tissues to elimination in expired air is described in Fig. 1. Carbon dioxide diffuses out of tissue cells to surrounding capillary claret (Fig. 1a). A small-scale proportion dissolves in claret plasma and is transported to the lungs unchanged.
Merely nigh diffuses into red cells where it combines with water to form carbonic acid. The acrid dissociates with product of hydrogen ions and bicarbonate. Hydrogen ions combine with deoxygenated hemoglobin (hemoglobin is acting equally a buffer hither), preventing a dangerous fall in cellular pH, and bicarbonate diffuses along a concentration gradient from ruby cell to plasma.
Thus virtually of the carbon dioxide produced in the tissues is transported to the lungs as bicarbonate in blood plasma.
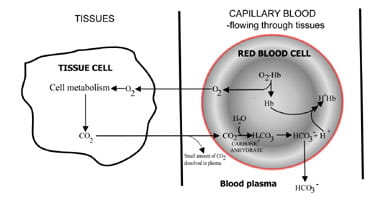
Fig. 1a. CO2 produced in tissues converted to bicarbonate for transport to lungs.
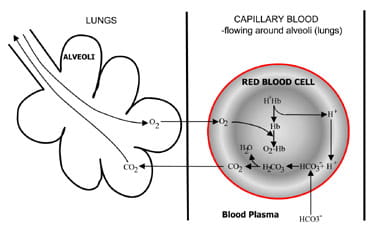
|
|
Fig. 1b. At the lungs bicarbonate converted back to CO2 and eliminated by the lungs.
At the alveoli in the lungs the procedure is reversed (Fig. 1b). Hydrogen ions are displaced from hemoglobin as information technology takes up oxygen from inspired air. The hydrogen ions are now buffered by bicarbonate which diffuses from plasma dorsum into reddish cell, and carbonic acrid is formed. Equally the concentration of this rises, it is converted to water and carbon dioxide. Finally, carbon dioxide diffuses down a concentration slope from carmine cell to alveoli for excretion in expired air.
Respiratory chemoreceptors in the brain stalk answer to changes in the concentration of carbon dioxide in blood, causing increased ventilation (breathing) if carbon dioxide concentration rises and decreased ventilation if carbon dioxide falls.
Kidneys and acrid-base residuum
Normal cellular metabolism results in continuous production of hydrogen ions. We take seen that by combining with these hydrogen ions, the bicarbonate buffer in blood minimizes their effect. Nonetheless, buffering is but useful in the short term, and ultimately hydrogen ions have to be removed from the body. Furthermore, it is important that the bicarbonate that is used to buffer hydrogen ions is continuously replaced.
These 2 tasks, elimination of hydrogen ions and regeneration of bicarbonate, are achieved by the kidneys. Renal tubule cells are rich in the enzyme carbonic anhydrase, which facilitates germination of carbonic acid from carbon dioxide and water. Carbonic acid dissociates to bicarbonate and hydrogen ions. The bicarbonate is reabsorbed into blood and the hydrogen ions pass into the lumen of the tubule and are eliminated from the trunk in urine.
This urinary elimination is dependent on the presence in urine of buffers, principally phosphate and ammonia ions.
DISTURBANCES OF ACID-BASE BALANCE
Near acid-base disturbances result from
- affliction or damage to organs (kidney, lungs, brain) whose normal office is necessary for acid-base homeostasis,
- disease which causes abnormally increased product of metabolic acids such that homeostatic mechanisms are overwhelmed
- medical intervention (east.chiliad. mechanical ventilation, some drugs)
Arterial blood gases are the blood test used to identify and monitor acid-base disturbances. Iii parameters measured during blood gas analysis, arterial blood pH (pH), partial pressure of carbon dioxide in arterial blood (pCO2(a)) and concentration of bicarbonate (HCO3 –) are of crucial importance (run into Tabular array I for reference (normal) range). Results of these three let nomenclature of acid-base of operations disturbance to 1 of four etiological categories:
- Respiratory acidosis
- Respiratory alkalosis
- Metabolic acidosis
- Metabolic alkalosis
| Adults | Neonates | |
| pH | vii.35-7.45 | 7.30-7.40 |
| pCO2 (kPa) | 4.seven-half-dozen.0 | 3.5-5.4 |
| Bicarbonate (mmol/L) | 22-28 | 15-25 |
TABLE I. Approximate reference (normal) ranges
To understand how the results of pH, pCO2(a) and bicarbonate are used to classify acid-base disturbances in this way, nosotros must return to the Henderson-Hasselbalch equation
pH = half-dozen.1 + log ([HCOiii –] / [H2CO3])
We mensurate pH and bicarbonate but not carbonic acrid (HiiCOiii). Yet, there is a relationship betwixt pCOtwo(a) and HtwoCO3 which allows restatement of the Henderson-Hasselbalch equation in terms of the three parameters (pH, pCOii(a) and bicarbonate) measured during blood gas analysis:
pH = half-dozen.1 + log ([HCO3 –] / (pCO2(a) × 0.23))
By removing all constants from this equation, the relationship between the three measured parameters tin be more only stated:
pH ∝ [HCO3 –] / pCO2(a)
This relationship, crucial for an understanding of all that follows apropos acrid base disturbances, states that arterial blood pH is proportional to the ratio of bicarbonate concentration to pCO2(a). It allows the post-obit deductions:
- pH remains normal and so long as the ratio [HCO3 –] : pCO2(a) remains normal
- pH increases (i.e. alkalosis occurs) if either [HCO3 –] increases or pCO2(a) decreases.
- pH decreases (i.east. acidosis occurs) if either [HCOthree –] decreases or pCO2(a) increases
- If both pCOtwo(a) and [HCO3 –] are increased by relatively the same corporeality, the ratio and therefore the pH are normal
- If both pCO2(a) and [HCOthree –] are decreased by relatively the same amount, the ratio and therefore the pH are normal.
Acid-base disturbances affect primarily either pCO2(a), in which instance it is called a respiratory disturbance, or [HCOiii –], in which case it is called a not-respiratory or metabolic disturbance:
- If the principal disturbance is a raised pCOii(a) (which causes acidosis – encounter higher up), the condition is called respiratory acidosis
- If the main disturbance is a reduced pCO2(a) (which causes alkalosis – see to a higher place), the condition is called respiratory alkalosis
- If the primary disturbance is associated with reduced bicarbonate (which results in acidosis – see above), the condition is chosen metabolic acidosis
- If the principal disturbance is associated with raised bicarbonate (which results in alkalosis – meet above), the status is chosen metabolic alkalosis
Causes of acid-base disturbances
Respiratory acidosis – (raised pCO2(a), reduced pH)
Respiratory acidosis is characterized by increased pCOii(a) due to inadequate alveolar ventilation (hypoventilation) and consequent reduced elimination of COii from the claret. Respiratory disease, such as bronchopneumonia, emphysema, asthma and chronic obstructive airways disease, may all be associated with hypoventilation sufficient to cause respiratory acidosis.
Some drugs (due east.1000. morphine and barbiturates) tin can cause respiratory acidosis by depressing the respiratory center in the brain. Damage or trauma to the chest wall and the musculature involved in the mechanics of respiration may reduce ventilation charge per unit. This explains the respiratory acidosis that tin complicate the course of diseases such as poliomyelitis, Guillain-Barre syndrome and recovery from severe breast trauma.
Respiratory alkalosis – (reduced pCOtwo(a), increased pH)
By contrast, respiratory alkalosis is characterized by decreased pCOii(a) due to excessive alveolar ventilation and resulting excessive elimination of CO2 from blood. Illness in which, due to reduced oxygen in claret (hypoxemia), the respiratory center is stimulated can consequence in respiratory alkalosis.
Examples hither include severe anemia, pulmonary embolism and adult respiratory syndrome. Hyperventilation sufficient to crusade respiratory alkalosis tin be a feature of anxiety attacks and response to severe pain. Ane of the less welcome properties of salicylate (aspirin) is its stimulatory effect on the respiratory center. This effect accounts for the respiratory alkalosis that occurs following salicylate overdose. Finally, overenthusiastic mechanical ventilation can cause respiratory alkalosis.
Metabolic acidosis – (decreased HCO3 –, decreased pH)
Reduced bicarbonate is always a feature of metabolic acidosis. This occurs for one of two reasons: increased utilize of bicarbonate in buffering an abnormal acrid load or increased losses of bicarbonate from the body. Diabetic ketoacidosis and lactic acidosis are two conditions characterized by overproduction of metabolic acids and consequent exhaustion of bicarbonate.
In the first example, abnormally high blood concentrations of keto-acids (b-hydroxybutyric acid and acetoacetic acid) reflect the severe metabolic derangements which result from insulin deficiency.
All cells produce lactic acrid if they are deficient of oxygen, so increased lactic acid production and resulting metabolic acidosis occur in any condition in which oxygen delivery to the tissues is severely compromised.
Examples include cardiac arrest and any condition associated with hypovolemic shock (e.g. massive fluid loss). The liver plays a major function in removing the small amount of lactic acid that is produced during normal jail cell metabolism, so that lactic acidosis can exist a feature of liver failure.
Abnormal loss of bicarbonate from the body can occur during severe diarrhea. If unchecked, this can lead to metabolic acidosis. Failure to regenerate bicarbonate and excrete hydrogen ions explains the metabolic acidosis that occurs in renal failure.
Metabolic alkalosis – (increased HCOiii – , increased pH)
Bicarbonate is always raised in metabolic alkalosis. Rarely, excessive administration of bicarbonate or ingestion of bicarbonate in antacid preparation can cause metabolic alkalosis, but this is usually transient. Abnormal loss of hydrogen ions from the torso tin can be the primary trouble. Bicarbonate which would otherwise be consumed in buffering these lost hydrogen ions consequently accumulates in blood. Gastric juice is acidic and gastric aspiration or whatsoever affliction process in which gastric contents are lost from the trunk represents a loss of hydrogen ions.
The projectile vomiting of gastric juice, for example, explains the metabolic alkalosis that tin occur in patients with pyloric stenosis. Severe potassium depletion tin cause metabolic alkalosis due to the reciprocal relationship between hydrogen and potassium ions.
Compensation – a result of acid-base of operations disturbance
It is vital for life that pH does not waiver too far from normal, and the trunk will always effort to render an abnormal pH towards normal when acid-base rest is disturbed. Bounty is the name given to this life-preserving procedure. To understand compensation, it is of import to recall that pH is governed by the ratio [HCOiii –] : pCOtwo(a). So long every bit the ratio is normal, pH will be normal.
Consider the patient with metabolic acidosis whose pH is low because bicarbonate [HCO3 –] is low. To recoup for the low [HCO3 –] and restore the all-important ratio towards normal the patient must lower his pCOtwo(a). Chemoreceptors in the respiratory heart of the brain reply to a ascension hydrogen ion concentration (low pH), causing increased ventilation (hyperventilation) and thereby increased elimination of carbon dioxide; the pCO2(a) falls and the ratio [HCO3 –] : pCO2(a) returns towards normal.
Bounty for metabolic alkalosis in which [HCO3 –] is high, by contrast, involves depression of respiration and thereby retentiveness of carbon dioxide and then that the pCOtwo(a) rises to friction match the increase in [HCO3 –]. However, depression of respiration has the unwelcome side effect of threatening acceptable oxygenation of tissues. For this reason respiratory bounty of metabolic alkalosis is limited.
Primary disturbances of pCOii(a) (respiratory acidosis and alkalosis) are compensated for by renal adjustments of hydrogen ion excretion which result in changes in [HCO3 –] that recoup appropriately for master modify in pCO2(a). Thus the renal compensation for respiratory acidosis (raised pCO2(a)) involves increased reabsorption of bicarbonate, and renal compensation for respiratory alkalosis (reduced pCOtwo(a)) involves reduced bicarbonate reabsorption.
The concept of acid-base balance during compensation is conveyed visually in Fig. 2. Table II summarizes the claret gas results that narrate all iv acid-base disturbances before and after compensation.
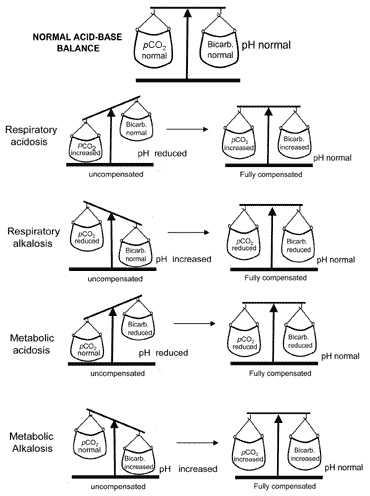
Fig. 2. The "acrid-base balance" : compensation restores normal pH
| Primary disturbance | ||||
| Respiratory acidosis primary increase in pCO2 | Repiratory alkalosis primary decrease in pCOii | Metabolic acidosis primary decrease in bicarb. | Metabolic alkalosis primary increase in bicarb. | |
| Some common causes | Emphysema COPD Pneumonia Low of repiratory centre | Hyper- ventilation Anxiety attacks Stimulation of brain respiratory center | Renal failure Diabetic ketoacidosis Circulatory failure - clinical shock (lactic acidosis) | Bicarbonate admini- stration Potassium depletion |
| Compen- satory mechanism | RENAL increase bicarbonate | RENAL subtract bicarbonate | RESPIRA- TORY decrease pCO2 | RESPIRA- TORY increase pCOtwo simply limited compen- sation in metabolic alkalosis |
| Initial blood gas results (uncompen- sated) | pH decreased pCO2 increased Bicarbonate normal | pH increased pCO2 decreased Bicarbonate normal | pH decreased pCO2 normal Bicarbonate decreased | pH increased pCOii normal Bicarbonate increased |
| Claret gas results after partial compen- sation | pH decreased but closer to normal pCO2 inreased Bicarbonate increased | pH increased only closer to normal pCO2 decreased Bicarbonate marginally decreased | pH decreased but closer to normal pCO2 marginally decreased Bicarbonate decreased | Limited compen- sation in metabolic alkalosis |
| Blood gas results afterwards total compen- sation | pH normal pCO2 increased Bicarbonate increased | pH normal pCOii decreased Bicarbonate decreased | pH normal pCO2 decreased Bicarbonate decreased | Limited compen- sation in metabolic alkalosis |
Impress friendly version of table, pdf.
TABLE II. Blood gas results in disturbances of acid-base balance
Respiratory compensation for a primary metabolic disturbance occurs much more rapidly than metabolic (renal) bounty for a master respiratory disturbance. In the second case, bounty occurs over days rather than hours.
If compensation results in return of pH to normal and so the patient is said to be fully compensated. Simply in many cases the compensation returns pH towards normal without really achieving normality; in such cases the patient is said to exist partially compensated.
For reasons described to a higher place, metabolic alkalosis is very rarely fully compensated.
Mixed acid-base disturbances
Information technology might be assumed from the in a higher place give-and-take that all patients with acrid-base disturbance suffer from only one of the iv categories of acid-base balance. This may well be the instance, but in detail circumstances patients tin present with more than than 1 disturbance.
For case, consider the patient with a chronic lung disease such as emphysema who has a long-standing partially compensated respiratory acidosis. If this patient were also a diabetic who had non taken his normal insulin dose and as a effect was in a state of diabetic ketoacidosis, claret gas results would reflect the combined effect of both respiratory acidosis and metabolic acidosis.
Such mixed acid-base disturbances are not infrequent and may exist difficult to unravel on the basis of arterial blood gas results alone.
Summary
The maintenance of normal blood pH involves several organ systems and depends on circulatory integrity. It is not surprising then that disturbance of acrid-base balance tin can complicate the grade of widely various diseases equally well equally trauma to many parts of the body. The torso has considerable ability to preserve blood pH, and disturbances usually imply either severe chronic disease or acute critical illness.
The results of arterial claret gas assay can identify acid–base of operations disturbance and provide valuable data as to its cause.
Some suggested farther reading
- Thomson WST, Adams JF, Cowan RA. Clinical acid-base balance. Oxford: Oxford Medical Publications 1997
- Harrison RA. Acid-base residuum. Respir Care Clin N. America 1995; 1,1: 7-21
- Woodrow P. Arterial blood gas analysis. Nursing Standard 2004; xviii,21: 45-52
- Sirker AA, Rhodes A, Gounds RM, Bennet ED. Acid-base physiology: the 'traditional' and the 'modern' approach. Anaesthesia 2002; 57: 348-56
Disclaimer
May incorporate information that is not supported by performance and intended utilize claims of Radiometer's products. Run into also Legal info.
Source: https://acutecaretesting.org/en/articles/an-introduction-to-acidbase-balance-in-health-and-disease
0 Response to "A Nurse Is Reviewing the Arteriol Blood Gas for a Client Who Has Respritory Acidosis"
Post a Comment